 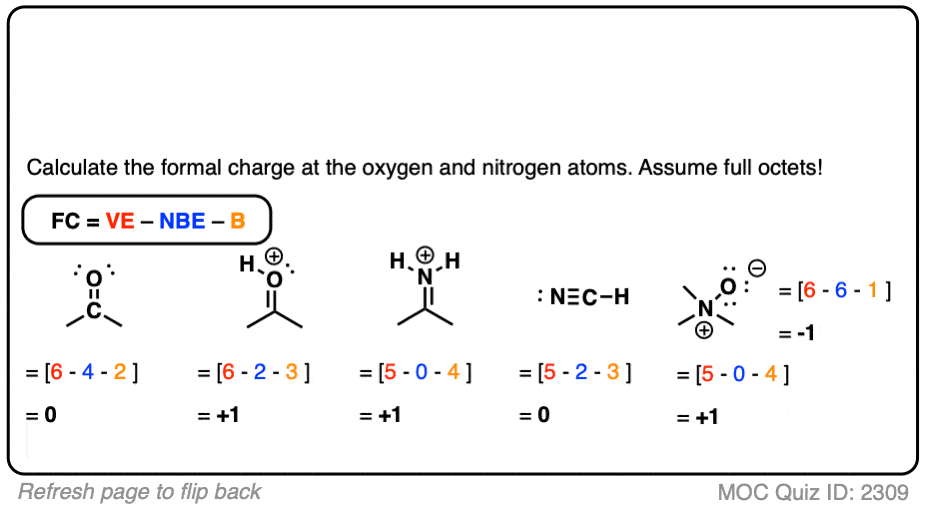
Three unshared pairs of electrons are written over the Oxygen atom of the molecule. The bonding pair electrons are shown with straight lines between two atoms.įor now in the lewis structure, total eighteen electrons (nine pairs of electrons) are involved in bond formation.Īs Hydrogen is a ‘group 1’ element it has no vacant orbital for the nonbonding electrons. Hydrogen and Oxygen atoms are placed around the Carbon atoms. Hydrogen has only one electron, so it can’t be the central atom.Ĭarbon becomes the central atom in the Acetone molecule. To be the central atom, at least it has to connect with two other atoms. Here Hydrogen is the less electronegative atom. In Acetone lewis structure, the arrangement of atoms occurs in a way that generally the atom with less electronegative character will be the central atom in a molecule. Arrangement of atoms in Acetone lewis structure Total electron pairs which participate in molecule formation= total outer shell electrons in the molecule ÷ 2. As there are 24 outer orbit electrons in an Acetone molecule. We can find how many pairs of electrons participate in the bonding in Acetone molecule by dividing the number 24 by 2 and the number will be 12. 
In a bond forming between two atoms next to each other, there two electrons (a pair of electron) are involved. Thus in a Acetone molecule the number of total electrons in the outer orbit of the atoms are 24.Īcetone lewis structure Number of electron pair calculation For that one Oxygen atom the total outer most shell electron number will be (6×1) = 6. For six Hydrogen atoms these electron number will be (1×6) = 6. 
In the Acetone molecule the total outer shell electrons for three Carbon atoms are (4×3) = 12. That of an Oxygen atom is six and for a Hydrogen atom is one. Hence the number of outer most shell electron in a Carbon atom is four. Electronic configuration of a Hydrogen atom: 1s1. Electronic configuration of an Oxygen atom: 1s2 2s2 2p4. Outer orbit electron calculationĮlectronic configuration of a Carbon atom: 1s2 2s2 2p2. Carbon is an element of ‘group 14’, Oxygen is an element of ‘group 16’ and Hydrogen is a ‘group 1’ element in Periodic table. To draw the lewis structure of Acetone, all the outer orbit electrons of these atoms are first calculated. So an acetone molecule contains three Carbon atoms, six hydrogen atoms and one oxygen atom. It is a smallest ketone molecule with molecular formula C3H6O or (CH3)2CO. Structural formula of Acetone moleculeĪcetone is a molecule in Carbonyl family. Or some of these electrons don’t take part in bond formation, remains as nonbonding electrons. These electrons may forms bond with the outer shell electrons of the atom next to it. The outer orbit electrons of a particular atom are shown by dot symbols around that particular atom. This article is discussing about the Acetone lewis structure and explanations.Īcetone Lewis structure is written with the atomic symbol of atoms in the molecule. Lewis dot structure gives an idea about outer most orbit electrons of an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
